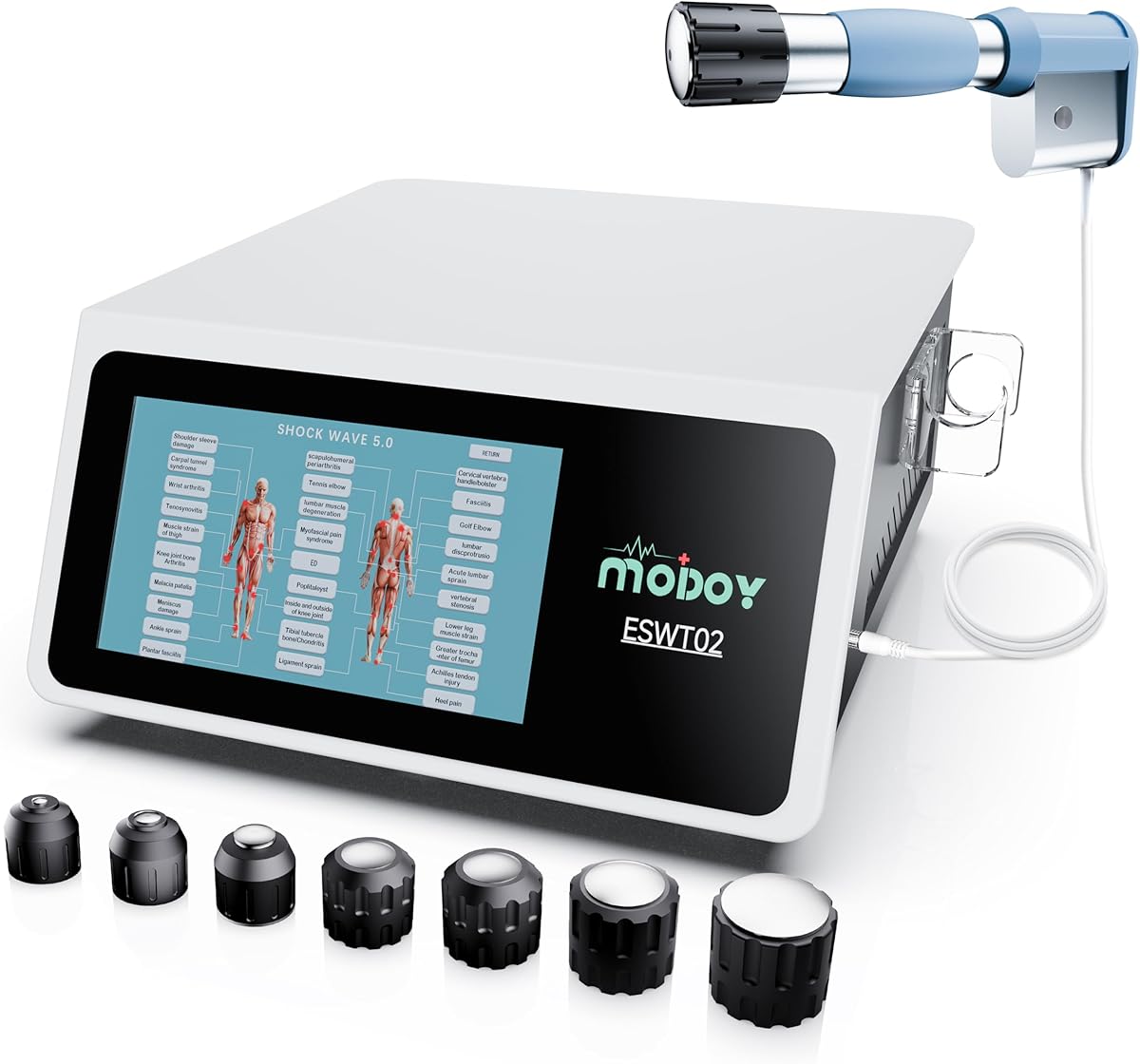
Evaluating Home Shockwave Therapy: What the MODOY 2026 Model Offers
The MODOY 2026 Shockwave Therapy Machine represents a specific class of home-use extracorporeal shock wave therapy (ESWT) devices. Unlike generic TENS units, this device generates focused acoustic waves intended to stimulate tissue repair, and its FSA/HSA eligibility indicates it meets certain medical device standards. Buyers should understand it's designed for chronic musculoskeletal conditions, not acute injuries.
Key Considerations Before Buying
- Clinical vs. Home Intensity: This device's energy output is calibrated for safe home use, which may differ from higher-intensity clinical ESWT machines used by physical therapists.
- Treatment Protocol Specificity: Effective use requires following specific protocols for duration, frequency, and applicator placement on areas like plantar fascia, shoulders, or elbows.
- FSA/HSA Eligibility Verification: While marketed as eligible, users must confirm with their plan administrator, as reimbursement often requires a Letter of Medical Necessity (LMN) for a diagnosed condition.
What Our Analysts Recommend
For ESWT devices, key quality indicators include clear energy level settings (measured in mJ/mm²), multiple applicator head sizes for different treatment areas, and a robust handpiece design. The MODOY's '2026' designation suggests a model iteration, implying potential refinements over earlier versions.
Muscle Stimulators & Accessories Market Context
Market Overview
The home-use ESWT market is rapidly expanding, with devices now bridging the gap between professional clinical equipment and over-the-counter pain relievers. Products like the MODOY 2026 compete directly with brands like Shockwave Medical and others offering portable, non-invasive alternatives.
Common Issues
Common user frustrations include unrealistic expectations for immediate pain relief (ESWT often requires multiple sessions over weeks), confusion between radial and focused shockwave technologies, and discomfort during application on bony prominences.
Quality Indicators
Look for devices with adjustable frequency (1-22Hz is typical), a pressure sensor in the applicator to ensure proper skin contact, and a manufacturer that provides clear, condition-specific treatment guidelines. Build quality of the handpiece and cable connections is critical for durability.
Review Authenticity Insights
Grade B Interpretation
A 'B' grade with a 15% estimated fake review rate suggests the review corpus is reasonably reliable but requires discerning reading. This level is common for emerging health tech products where enthusiastic early adopters and skeptical users both leave genuine feedback.
Trust Recommendation
Focus on the verified purchase reviews that describe specific, multi-week usage patterns for conditions like plantar fasciitis or tennis elbow. Be cautious of reviews that sound like promotional copy or mention free products, as these may constitute the estimated inauthentic portion.
Tips for Reading Reviews
Prioritize reviews that discuss the learning curve for proper use, the consistency of results over a month, and any technical support experiences. For this device, pay special attention to comments about battery life per charge and the ergonomics of the handpiece during a 15-minute session.
Expert Perspective
The MODOY 2026's adjusted rating of 4.10/5 from verified purchasers indicates a generally positive reception among buyers who have realistic expectations for home-based ESWT. The fact that all reviews are verified purchases significantly bolsters the credibility of the feedback, suggesting users are genuinely investing in a solution for chronic pain. The product's positioning for both home and clinical use suggests it aims for a higher power output than basic consumer models, which is a key differentiator.
Purchase Considerations
Weigh the upfront cost against the potential for repeated professional therapy sessions. Consider if your specific condition (e.g., calcific tendonitis vs. general muscle soreness) is clinically supported for ESWT treatment. Assess your comfort with self-administering a device that requires precise placement and consistent treatment schedules.
Comparing Alternatives
Shoppers should compare the MODOY's specifications, particularly its energy density range and pulse frequency settings, against other FSA/HSA-eligible portable ESWT devices.